Pharma organizations are slowly but steadily moving into digitization transformation of their business processes [1]. Their corresponding IT organizations are following that trend by supplying the digital technology driving the transformation. The technology itself is provided by the technology companies that have switched their operational models to the agile, quite some time ago to better meet the technology development demands and address efficiencies of their processes. However, it is typically not so with Pharma IT organizations implementing systems into their processes. Agile GxP systems implementation is seen as the compliant solution to provisioning systems in Pharma. As the Pharma industry, in general, is a science-driven industry designed around narrow scientific domains it remains relatively traditional in its approach and with reduced ability to iterate quickly [2].
Most of the GxP implementation projects are still executed using traditional V-model. To meet the digitalization demands these IT organizations would need to bring in more of the agile principles into their implementation of GxP systems and system updates.
Traditionally, implementation of GxP systems was focused around providing the full set of capabilities, and thereby full value, in the single implementation effort. Such an effort was, therefore, quite an extensive, long, and risky activity. Often, by the time the system is implemented the original requirements were no longer fully applicable and the system would need to undergo a change through the change request mechanism, taking a long road to implementing that change. It is equivalent to turning a huge ship in narrow spaces – quite a difficult and risky endeavor. To meet today’s requirements for flexibility and agility of the systems supporting the flexible and agile processes, the implementation of GxP systems must be made agile and adaptable to delivering initial value as quickly as possible and then strategically dealing with additional values through the agile approach – delivering constant value stream. This approach is equivalent to moving the load of the ‘huge ship’ using smaller speedboats delivering movement agility and rapid change of pace and direction that modern organizations need.
The organizations do not launch agile transformations to remove pain points, but because they want to build the foundation for the company to compete in the future.
Agile Organization
Agile organizations are built around highly flexible and well-networked teams that are equipped to operate in accelerated learning and decision-making modes. They can quickly redirect their people and priorities toward value-creating opportunities. Within agile organizations, teams are given access to the information so that they can form decisions and act in the direction of the agreed common purpose. Such teams can maintain stability and efficiency in their service delivery by utilizing empowerment, velocity, and adaptability. The key to building an agile organization is in the ability to form a common purpose and agile culture.
One of the key ingredients in building an agile culture is the ability to form dynamic, collaborative, and interconnected teams. In a recent article by McKinsey organization about leadership in crisis (related to COVID-19 and future challenges), the authors are discussing steps required to build a network of teams capable to respond to changing environments with urgency [3]. These networked teams will be able to make faster and better decisions in the environment with rapidly shifting priorities and challenges. To create such highly dynamic and team-lead transformations, one would often begin with a central team operating in connection to a few response teams. The team evolutionary process would continue with hub and spoke configuration as the other teams would join the organization and result in a fully networked organization of teams, capable to connect and collaborate directly with one another.
Having agile and networked teams within a highly dynamic Clinical Development organization, one can embrace digital transformations of the traditional processes, such as protocol development, patient engagement, or clinical supply, etc. The recent concept of a “digital clinical trial” involves leveraging digital technology to improve patients’ access, engagement, protocol assessments, interventions, and is considered to be the gateway to transform clinical trials and to lower their cost [4].
However, to enable that transformation to happen, the development organization must also rapidly respond with a matching Quality organization capable to follow the dynamics with an appropriate agile GxP systems provisioning. Without the agile GxP organization, the digital transformation in any part in the clinical development organization is bound to slow or even stall bringing the entire business case for the transformation in jeopardy. In that undesirable case, the GxP systems provisioning would become a bottleneck for the entire organization.
Transforming to Agile GxP Systems Implementation Model
Agile transformation is not only an all-embracing of the entire GxP delivery organization but is also iterative, where the target state of the organization is constantly refined by the lessons learned from the pilots executed in earlier stages of the transformation. Essential to the agile transformation success is the strong alignment and support from the top management. A typical transformation starts as a pilot stage, with learnings converted into a better determination of the future organization state followed by scale and improve stage where agile methods are rolled out broadly within the organization.
The most common current state of the GxP project delivery organization across the industry is the waterfall, i.e. V-model for implementing GxP systems. The V-model is typically implemented as the linear model following the so-called V-cycle of URS->FRS->DS->Implementation->IQ->OQ->PQ. This model is very conservative and the change in requirements during the project lifecycle should be avoided as due to the waterfall nature it inevitably leads to delay and increase of the project costs. The organization must evolve from the current V-model into an agile model but at the same time must ensure that the future state maintains full regulatory compliance. To do that, the organization needs to have senior management support and computerized systems and tools in place to be able to make the required leap.
Therefore, in the pilot stage, the top management support and aspiration need to be established with definitions of the GxP project delivery organization’s value streams, design of the organization structure, and identification of the agile teams. The agility enablers in terms of processes, people, and technology need to be specified as those enablers provide the backbone of the transformation itself. With these essential elements of the operating model, a roadmap outlining the implementation approach provides a complete blueprint for the GxP delivery organization with a clear vision and the operating model design.
In a GxP Project Delivery Organization, pilots could address a specific project in the organization and create learnings that will be applied to the agile ways of working to the broader GxP delivery organization through the “scale and improve” efforts.
Transformation Iterative Approach
Since it would be quite safe to assume that in the Pharma industry the organizations are not of the borne-agile type as many in the technology sector, they must undergo a transformation to embrace enterprise agility. A recent McKinsey article on “the journey to an agile organization” [5], outlines broadly two components iterative approach to continually test, learn, and correct the road to the agile organization:
- Aspire, design and pilot component
- Scale and improve component
The first component “Aspire, design and pilot” is a kind of a grassroots component, that plants the seeds for the second component “Scale and improve” which nurtures those grassroots and grows the agile organization.
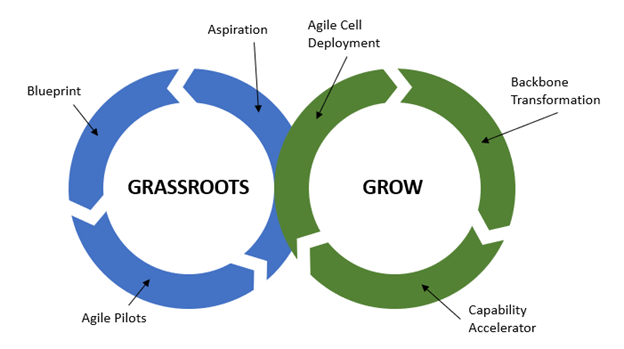
In the grassroots phase, we agree on the vision of the future state of the agile organization and develop the blueprint which we are continually testing, learning, and correcting through agile pilots. The nurture and grow phase is all about rolling out agile cells within the organization, transforming the backbone, and systematically build the organization’s capabilities.
In the GxP validation organization, we can start with building the grassroots around projects that are potentially well suited for the pilot. By that, we mean projects that involve business processes and technologies that offer possibilities to define the value delivery in waves that make the most sense to the end-user. In this determination, interaction with the customers, in this case, end-users, is essential to define the value waves and benefits expected after each wave is delivered. In this way, we can blueprint the agile teams and methodologies to execute the projects with agility and deliver the expected values to the end-user as quickly as possible to keep the dynamic of the business process to its final state.
When more of the projects are included in the agile operation, the original blueprint would be refined and the agility backbone of the delivery organization will grow to include more of the agile teams until eventually all of the GxP project delivery organization is transformed.
Of course, this entire process is not simple and it requires a great deal of aspiration, energy, communication, and mindset change to make it happen, but once it is in place a whole new set of capabilities emerge that propel the organization higher in the competitive space.
We at intilaris have spent decades in the traditional GxP project delivery operation manifested in the V-model but have recognized the need to bring agility to the GxP systems implementation to be able to follow the pace of digitalization. Therefore, we have invested in building the skills required to support our customers on their GxP project delivery and transformation to agile.
[1] Sandle, Tim. (2019). Digital Transformation of Pharmaceuticals and Healthcare.
[2] https://hbr.org/2019/11/why-science-driven-companies-should-use-agile
[4] Inan, O.T., Tenaerts, P., Prindiville, S.A. et al. Digitizing clinical trials. npj Digit. Med. 3, 101 (2020). https://doi.org/10.1038/s41746-020-0302-y


